- Home Page
- Company Profile
-
Our Products
- Vaccine Products
- Inactivated Influenza Vaccine IP
- 5 ml Inactivated Influenza Vaccine IP
- Meningococcal Polysaccharide Diphtheria Toxoid Conjugate Vaccine
- Measles Mumps And Rubella Vaccine IP
- Typhoid Vi Conjugate Vaccine IP
- Varicella Vaccine
- Vaxiflu-4 Inactivated Influenza Vaccine IP
- Menactra Conjugate Vaccine
- Typbar TCV Typhoid Vaccine
- Influvac Influnac Inactivated Influenza Vaccine
- VaxiFlu Inactivated Influenza Vaccine
- Tresivac PFS Measles Mumps And Rubella Vaccine
- Variped Varicella Vaccine
- Rabies Vaccine
- Anti Rabies Injection
- Abhayrab Rabies Vaccine
- Xprab Rabies Vaccine
- Onco Products
- 400 MG Bevacizumab Injection
- 1 MG Anastazole Tablets
- 100 MG Bevacizumab Solution For Injection
- 400 MG Bevacizumab Concentrate For Solution For Infusion
- Leucovorin Calcium Tablets USP
- 100 MG Cytarabine Injection BP
- L-Asparaginase For Injection
- 5 ML Methotrexate Injection IP
- Silodosin Capsules
- 1.4 GM Gemcitabine Injection IP
- 1 GM Gemcitabine Injection IP
- Bevacizumab Solution For Infusion
- 150 MG Trastuzumab Lyophilized Powder For Concentrate For Solution For Infusion
- 300 MG Filgrastim Injection IP
- 10 ML Ferric Carboxymaltose Injection
- 500 MG Abiraterone Acetate Tablets IP
- 100 MG Rituximab Solution For Infusion
- 5 ML Fulvestrant Injection
- 260 MG Paclitaxel Injection IP
- 150 MG Trastuzumab For Injection
- Rituximab Solution For Infusion
- Rituximab Concentrate For Solution For Infusion
- 60 MG Denosumab Solution For Injection PFS
- 50 ML Oxaliplatin Injection IP
- 11.25 MG Triptorelin Powder For Injection
- 500 MG Pemetrexed Injection IP
- 250 MG Abiraterone Acetate Tablets
- 10 ML Rituximab Solution For Infusion
- Mycobacterium W Injection
- 100 MG Rituximab Injection
- 1 MG Vincristine Sulphate Injection USP
- 50 MG Doxorubicin Hydrochloride Injection IP
- Exquisite Pharma Products
- 100 ML Ambroxol Hydrochloride Terbutaline Sulphate Guaiphensin And Menthol Syrup
- Luliconazole Soap
- Itraconazole Capsules BP
- Dextrose Zinc Vitamin C Lactobacillus Electrolytes Energy Drink
- 75 GM Luliconazole Absorbent Dusting Powder
- 15 ML Luliconazole Lotion 1% W-V
- 10 GM Cefixime Oral Suspension IP
- 100 ML Levosulbutamol Sulphate Ambroxol Hydrochloride And Guaiphensin Syrup
- 60 ML Montelukast Sodium And Levocertirizine Dihydrochloride Suspension
- Itraconazole Capsules
- 500 MG Azithromycin Tablets IP
- 30 ML Cefpodoxime Proxetil Oral Suspension IP
- 100 ML HBR Phenylephrine HCL And Chlorpheniramine Maleate Syrup
- 130 MG Itraconazole Capsules
- Itrazit Itraconazole Capsules
- 225 ML Lycopene Multivitmin And Multiminerals Syrup
- Cefixime And Lactic Acid Bacillus Tablets
- Pantoprazole Sodium And Domperidone Capsules IP
- Amoxycillin And Potassium Clavulanate Tablets IP
- Amoxycillin Potassium Clavulanate And Lactic Acid Bacillus Tablets
- Curcumin C3 Complex Bioperine Vitamin B12 Vitamin D2 And Multivitamin Tablets
- Diclofenac Potassium And Paracetamol Tablets
- Ginseng Multivitmins Multiminerals And Nutrients Tablets
- Aceclofenac Paracetamol And Serratiopeptidase Tablets
- Papaya Leaf Ext And Guduchi Roof Ext Tablets
- Levocertirizine Hydrochloride And Montelukast Sodium Tablets IP
- Tocopherols L-Carnitine Tartrate Vitamin D Tablet
- Calcium Magnesium Sulphate Zinc Sulphate Folic Acid Vitamin D Tablet
- Pantoprazole Sodium Gastro-Resistant Tablets IP
- Cefpodoxime Proxetil Dispersible Tablets
- Ketorolac Tromethamine Dispersible Tablets
- Coenzyme Q10 Berberis Omega 3 Fatty Acid L-Carnitine L-Tartrate Tablets
- Coenzyme Q10 Berberis Omega 3 Fatty Acid L-Carnitine L-Arginine Tablets
- 20 MG Rabeprazole Gastro-Resistant Tablets IP
- Terbinafine Tablets IP
- Major Pharma Brands
- Oncology Medicines
- 100mg Bevarest Bevacizumab Injection
- Bionase 10k Injection
- 7.5mg Eligard Leuprolide Acetate Injectable Suspension
- 100mg Enfiera Rituximab Injection
- 1.4g Citafine Gemcitabine Injection
- 100mg Toritz Rituximab Injection
- 250mg Zecyte Abiraterone Acetate Tablets
- 150mg Hertraz Trastuzumab Injection
- 500mg Mabtas Rituximab Concentrate Solution For Infusion
- 50mg Zubidox Doxorubicin Hydrochloride Injection
- 500mg Mytera Abiraterone Acetate Tablets
- Encicarb 500mg Ferric Carboxymaltose Injection
- 500mg Pemetero Pemetrexed Injection
- 1g Citafine Gemcitabine Injection
- 300mcg Emgrast Filgrastim Injection
- 150mg Eleftha Infusion
- Silostar D Silodosin Dutas teride Capsules
- Sepsivac Mycobacterium Injection
- 1mg Arimidex Anastrazole Tablets
- Abevmy Bevacizumab 400 Mg Injection
- 400mg Cizumab Bevacizumab Infusion
- Bevatas 400 Mg Injection
- 1mg Vinlon Vincristine Sulphate Injection
- 100mg Rituxem Rituximab Injection
- 250mg Fulvira Fulvestrant Injection
- 11.25mg Pamorelin Triptorelin Injection
- Biotrexate 500mg Methotrexate Injection
- 260mg Gros Paclitaxel Injection
- Pegasta 6 Mg Injection
- Biobin 100mg Cytarabine Injection
- Biovorin 15mg Tablet
- 100mg Maball Rituximab Injection
- 100mg Oxa Oxaliplatin Injection
- Hepatitis B Vaccine
- Abraxane Nab-Paclitaxel Injection
- Pemetrexed Injection 500 Mg
- Pemetrexed For Injection
- Axzyb 5mg Axitinib Tablet
- Silostar D Silodosin Dutasteride Capsules
- Aprepitant Capsules 125 Mg
- Tegonat 15 CAP 7 s blister STRIP
- Cyclobenzabrine Tablet
- Pharmaceutical Tablets
- Ezarvita Ginseng Multivitamins Multimineral Nutrients Tablets
- Aceclofenac Paracetamol Serratiopeptidase Tablets
- 50mg Q Zit Tablets
- Curcu Zit Tablets
- 100mg Q Zit Tablets
- Terbozit Terbinafine Tablets
- Osteozit Tablets
- Pythoros DT Ketorolac Tromethamine Dispersible Tablets
- Dp Zit Diclofenac Potassium Paracetamol Tablets
- 500mg Azitox Azithromycin Tablets
- 625mg Clavozit LB Tablets
- 375mg Amoxicillin Potassium Clavulanate Tablets
- Cifixit LB Cefixime Lactic Acid Bacillus Tablets
- Jardiance Tablet 10 Mg
- Levocetirizine Hcl Montelukast Sodium Tablets
- 200mg Podzit Cefpodoxime Proxetil Dispersible Tablets
- Osteozit LC Tablets
- 20mg Rabozit Rabeprazole Gastro Resistant Tablets
- Pantozit Pantaprozole Sodium Gastro Resistant Tablets
- Cyclopam Tablets
- Trajenta 5 Mg
- Glyxambi 25mg 5mg Tablet
- Combiflam Tablet
- Cardace 5
- Methimez 5 Mg
- Mesacol 800 Mg Tablets
- 20mg Rosuvas Rosuvastatin Tablets IP
- Cozi QL Ubiquinol Acetate L Carnitine Astaxanthin Lycopene & Zinc Tablets
- Pantoprazole Sulphide Tablets
- 10mg Everolimus Tablets
- 10mg Jardiance Empagliflozin Tablets
- 5 Mg Ulipristal Acetate Tablets
- Nurokart L Methylfolate Calcium Mecobalamin Pyridoxal 5 Phosphate Tablets
- Vitamin B12 Mouth Dissolving Tablets
- Calcium Carbonate And Vitamin D3 Tablets
- Acetylcysteine Effervescent Tablets
- Folic Acid Tablet
- L Ornithine L Aspartate Silymarin Selenium Alpha Lipoic Acid Thiamine Tablets
- Sumo Cold Paracetamol Phenylephrine Hydrochloride Caffeine Diphenhydramine Hydrochloride Tablets
- 90mg Axcer Ticagrelor Tablets IP
- 10mg Storvas Atorvastatin Tablets IP
- 1mg Arimidex Anastrazole Tablets
- 5mg Storvas Atorvastatin Tablets IP
- Regen Flex C2 Rosehip Extract Hyaluronic Acid And Curcuma Tablets
- Vitamin C And Zinc Tablets
- Myo Inositol N Acetylcysteine L Arginine Vitamin D Tablets
- Methylcobalamin Pyridoxine HCL Folic Acid Tablets
- Mubivit SL Silymarin L Ornithine L Aspartate Inositol And B Complex
- 100mg Panthocal Calcium Pantothenate Tablets
- AFANAT 20 28 s BT BOTTLE
- 10mg Rosuvas Rosuvastatin Tablets IP
- Lamino Essential Amino Acids Tablets
- 2500mcg Biotin Mouth Dissolving Tablets
- Mec MD L Methylfolate VitaminB12 Pyridoxal 5 Phosphate & Vitamin D3 Sublingual Tablets
- L-Ornithine L-Aspartate Pancreatin Silymarin Tablets
- APNAT 60MG TABS 120 S BOTTLE
- Erlonat Erlotinib 150 Mg Tablets
- Geffy 250 Mg Tablet
- 1120mg Argocal Calcium Aspartate Anhydrous Tablets
- Jalra 50500 Mg Tablet
- Pyridoxine Hydrochloride Tablets
- 50mg Jalra Vildagliptin Tablets
- Cissus Quandragularis Tablets
- L Methylfolate Calcium Tablet
- Glucosamine HCI Methyl Sulphonyl Methane Boswellia Serrata Curcumin Sodium Hyaluronate Vitamin D
- 10mg Lyvelsa Finerenone Fim Coated Tablets
- 20mg Lyvelsa Finerenone Film Coated Tablets
- Garcinia Cambogia Fruit Rind Green Tea Leaf White Kidney Bean L Carnitine L Tartrate Tablet
- 1mg Tata Ferrous Ascorbate And Folic Acid Tablets IP
- Follihair Nutraceutical Amino Acids Vitamins Mineral Biotin Melatonin And Botanical Extracts Tablets
- Renolyl N Acetylcysteine And Taurine Tablets
- Cissus Quadrangularis Linn Extract With Boswellia Serrata Tablet
- Ferrous Ascorbate Folic Acid Zinc Vitamin D3 And Cyanocobalamin Tablets
- 0.5mg Methylcobalamin L Methylfolate Calcium And Pyridoxal 5 Phosphate Tablet
- Benfotiamine Alpha Lipoic Acid Inositol Mecobalamin Folic Acid Chromium And Selenium Tablets
- Fericip XT Ferrous Ascorbate And Folic Acid Tablets IP
- Feritus XT Ferrous Ascorbate And Folic Acid Tablets
- Gudplat Papaya Leaf Tablets
- Diasum GM Diacerein Glucosamine Sulphate Methyl Sulphonyl Methane Tablets
- Phenoxymethyl Penicillin Tablet
- Ranitidine Hydrochloride Tablets
- Diclofenac Sodium Tablets
- Lapatinib Tablet Ip 250mg Tylidys
- Emrok O 500 Mg Tablets
- Cacit 500mg Tablets
- Hvtek Plus Ivermectin
- XPREZA 300mg TAB 14 s BT BOTTLE
- Ibrunat 140 Mg
- L Glutathione Capsules
- DASANAT 50 60 S BT BOTTLE
- Lorviqua Lorlatinib Lorbrena
- Cissus Quadrangularis Linn Extract Tablet
- HERDUO 250mg 30 TAB S BOTTLE
- Cyclopam Tablets 20gm
- Ivermectin 12mg Tablets
- Iverheal 12mg USP Ivermectin Tablets
- Ivermectin Albendazole Tablets
- Ivertoro 12 Mg Tablets Ivermectin Tablets USP
- Q ZIT 100 MG
- Ivermectin Tablets 20mg
- Ivermectol 6mg Tablets
- Iverlast Ivermectin Tablets 12 Mg
- L Glutathione Hyaluronic Acid Vitamin C And Biotin Tablets
- Moringa Leaf Tablets
- 5mg Trajenta Linagliptin Tablets
- BRACANAT 150 30 TABLETS BOTTLE
- Bfol D3 Methylcobalamin Folic Acid And Vitamin D3 Sublingual Tablets
- XTANE 25 MG TABLETS 30 S BT BOTTLE
- XPREZA 200mg TAB 14'S BT BOTTLE
- Combiflam Tablet 20TAB
- Febrex Plus Paracetamol Chlorpheniramine Maleate Phenylephrine Hydrochloride Tablets (20x10 Box)
- SORAFENAT 200 Mg TAB 60 S BT BOTTLE
- Calcium Citrate Malate Zinc Oxide Magnesium Hydroxide Vitamin D3 & K2 Tablets (2x15)
- Dasatinib Invista 50mg, 100mg, 70mg
- Calcium Pantothenate, Carnitine, L-Ornithine L-Aspartate, Lecithin, NAC, Silymarin Tablets (500 Mg)
- 25mg Jardiance Empagliflozin Tablets
- Lycopene Antioxidants Essentials Amino Acids Multivitamins And Multiminerals Tablets
- Iverheal Ivermectin Tablets 12mg
- Ivermectin 40mg Tablets
- Ivermectin Tablets 24 Mg
- 200mg Gesteofit SR Progesterone Sustained Release Tablets
- Albendazole &Amp; Ivermectin Tablets
- Ivermectin 3 Mg Tablets
- Anastrozole Tablets 1 Mg
- Methimez 5 Mg
- 5mg Rosuvas Rosuvastatin Tablets IP
- Silymarin L-Ornithine L-Aspartate Inositol B Complex Tablets 150mg
- 50mg Istavel Sitagliptin Phosphate Tablets IP
- 5000mcg Biotin Sublingual Tablets
- 20mg Storvas Atorvastatin Tablets IP
- Cabotib Cabozantinib 204060 Mg Tablet
- Soframycin Skin Cream
- Nuparp 250mg 300mg Rucaparib Tablet
- Calcium Citrate, Magnesium Hydroxide, Zinc & Vitamin D3 Tablets
- Phenoxymethyl Penicillin Tablet 250 Mg
- NT Liver Silymarin L Ornithine L Aspartate Multivitamin Multimineral Antioxidant Tablet
- Lenalidomide Capsule 25mg
- Palbociclib 125 Mg
- Calizon D Calcium Carbonate And Vitamin D3 Tablets IP
- XPREZA 200mg TAB 14 S BT BOTTLE
- 1000mg Micronized L Arginine Tablet
- ERLONAT IP 100 MG 30 s BT BOTTLE
- Glucosamine And Chondroitin Tablets
- Distamine 500 Mg Tablets
- Allegra 180mg Tablet
- Ferrous Fumarate Tablet
- Silymarin L Carnitine L Ornithine Co Enzyme Q10 With Antioxidant And Micronutrients Capsules
- AFANAT 40 28 S BT BOTTLE
- Lenalid Lenalidomide Capsule 15 MG
- Terbutaline Sulphate Bromhexine Tablets
- Viraday Tablets 30 Tab
- Zoldonat 4mg Injection
- Mubivit SL Silymarin L Ornithine L Aspartate Inositol And B Complex Tablets
- Appavisc Pfs 3Ml
- 250mg Osteozit Tablets
- Anti Oxidant Capsules
- 4 Mg Zoledronic Acid Injection
- Vinorelbine Injection Usp
- L Aginine HCI Chasteberry N Acetyl L Cysteine Inositol Omega 3 Acid Co Enzyme Grape Seed Ext Zinc
- Qubidvi DHA Docosahexaenoic Acid Folic Acid & Vitamin B12 Softgel Capsules
- Irinotecan Hcl Injection
- Calciva Calcitriol Calcium Citrate Zinc Sulphate Monohydrate & Magnesium Oxide Softgel Capsules
- Live Freeze Dried Lactic Acid Bacteria And Bifidobacteria Capsules
- Evermil 5mg, 10mg Everolimus Tablets
- Larginine And Proanthocyanidin Granules
- Xefta 250 Mg
- Relugonat 120mg 30's BT BOTTLE
- Pharmaceutical Capsules
- 130mg Itrazit SB Itraconazole Capsules
- 130mg Itrazit SB Capsules
- Pantoprazole Sodium Domperidone Capsules
- 200mg Itraconazole Capsules
- LENALID 10 MG BT 30 S BOTTLE
- Undenatured Collagen Type 2
- Coenzyme Q10 Capsules
- 75mg Pradaxa Capsule
- 12 5mg Sunitinib Malate Capsules
- SUTINAT 25 28 CAPSULES BOTTLE
- Benfowel Benfotiamine Methylcobalamin Alpha Lipoic Acid Folic Acid Soft Gel Capsules (100mg Box)
- Ginseng Multivitamins Multiminerals Capsules
- Co Enzyme Q10 Capsule
- KNILONAT 200MG CAPS 10 s BS STRIP
- Nicotinamide 180 cap
- Ginkgo Biloba Extract Capsules
- Vitamin B6 Capsules
- 140mg Imbruvica Capsules
- Crizalk 250 Mg Capsule
- LENALID 15MG CAPS 30 S BT BOTTLE
- Undenatured Collagen Type 2 Capsules
- Pradaxa 110 Mg Dabigatran Etexilate Capsules
- VEENAT 100 MG CAP s 10 s BS STRIP
- Boswellia Serrata Extract Capsule
- Pradaxa 150 Hard Capsules
- PALENO 125mg Caps 21 s BOTTLE
- Amino Acid Capsules
- Methyl Cobalamin Capsule
- Dortas T Eye Drops
- 200mg Itrazit Itraconazole Capsules
- Febrex Plus Paracetamol Chlorpheniramine Maleate And Phenylephrine Hydrochloride Tablets
- Ginseng Multivitamins Multiminerals Capsules
- Pre Biotic And Pro Biotic Capsule
- Methyl Cobalamin Tablet
- 500mg Omega 3 Fatty Acid Soft Gel Capsule
- Docetaxel Injection 20 Mg
- CAZANAT 60 mg Tabs. 30 s BT BOTTLE
- Mesacol Suppository 1 g
- Niftran 100mg Capsule
- Benfowel Benfotiamine Methylcobalamin Alpha Lipoic Acid & Folic Acid Soft Gel Capsules
- Calcium Citrate Malate Zinc Oxide Magnesiun Hydroxide Vitamin D3 & Vitamin K2 7 Tablets
- Calcium Pantothenate + Carnitine + L-Ornithine - L-Aspartate + Lecithin + NAC + Silymarin Tablets
- L Carnitine L Tartrate Tablets
- 50 500 Istamet Sitagliptin Phosphate And Metformin Hydrochloride Tablets
- Vitamin B Complex And Vitamin C Capsules
- Zincovil Multivitamins Multiminerals And Natural Extract Tablets
- Calcium Citrate Malate Tablets
- Alpha Lipoic Acid Capsule
- Daunorubicin Injection 20 Mg
- Lenangio Capsules 10 mg
- Frulacto Pre And Probiotic Capsule
- Pyrinus D3 L Methylfolate Pyridoxal 5 Phosphate Methylcobalamine & Vitamin D3 Tablets
- 100mg Istavel Sitagliptin Phosphate Tablets IP
- Naproxen 500 Mg Tablet
- ERLONAT 150 MG TAB S 30 S BT STRIP
- Zincovit Multivitamin Multimineral And Grape Seed Extract Tablets
- Ostewin D3 Calcium Citrate Magnesium Hydroxide Zinc And Vitamin D3 Tablets
- Xtrant Capsules 140mg
- D Chiro Inositol Tablet
- Lomustine 40 Mg Capsule
- Pharmaceutical Syrup
- Pharmaceutical Injection
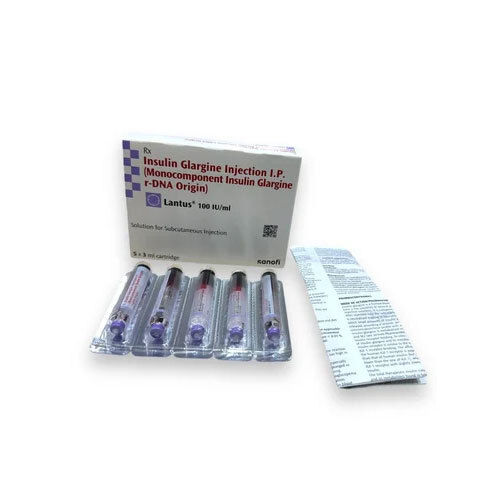
- Lantus Cartridge
- Hynidase Inj
- Dengue Ns1 Ag Rapid Test Kit
- CARFILNAT INJ 60MG VIAL
- NATDOX LP 20MG 10ML VIAL
- Gerta Ertapenem Injection
- Napro Tax 100 Mg Injection
- Infimab Infliximab 100 Mg Injection
- XPREZA INJECTION 100 MGVAIL
- Anti-D Immunoglobulin Injection
- Mounjaro 2.5 Mg Injection
- Hynidase Inj 1500 lu
- Lantus Cartridge
- BORTENAT 3.5MG INJ
- 1% Oculan Lignocaine Hydrochloride Injection IP
- Ferric Carboxymaltose Injection 50 MgMl
- 100mg Hemfer Iron Sucrose Injection USP
- 50 IU Humalog Mix Pen Insulin Lispro And Insulin Lispro Protamine Injection
- Spectinomycin Injection 1 G
- Melphalan Injection 50mg
- Oxaliplatin 100 Mg Injection
- XPREZA INJECTION 100 MG VAIL
- Bevatas 400 Mg Infusion
- Bryxta 400mg Bevacizumab Injection
- Paclitaxel 300mg Injection
- Vaxirab N 1 Ml
- Atsure Injection 1 G
- Piperacillin Tazobactam Injection
- 400mg Darzalex Injection
- Bortezomib Injection Ip
- FULVESER 250MG INJ VIAL
- Ifosfamide Mesna Injection 1 G
- Pemnat Injection 500 Mg
- Avastimab 100mg &Amp 400mg Bevacizumab Injection
- Bortecad 2.5mg Bortezomib Injection
- FULVENAT 250MG INJ - VIAL
- Vaxirab N 1 Ml
- Humapen Savvio 0.5 Ml
- 500mg Hemfer FCM Ferric Carboxymaltose Injection
- Azacitidine 100 Mg Injection
- Gemcite Injection 1000 Mg
- Bleomycin For Injection
- Topotecan Injection 4mg
- 100 IU Humalog Mix Pen Insulin Lispro And Insulin Lispro Protamine Injection
- Deghor 80mg Degarelix For Injection
- Danyelza Injection 40 Mg
- ZOLDONAT 4 MG INJ VIAL
- 100 IU Humalox Mix 25 Insulin Lispro And Insulin Lispro Protamine Injection
- KABANAT INJECTION 60MG1.5ML
- Medicated Soap
- Dusting Powder
- Pharmaceutical Lotion
- Pharmaceutical Gel
- Energy Drink Powder
- Anti Cancer Medicines
- Mounjaro 2.5 Mg
- Fruquintinib Capsules 5mg
- DASANAT 20 60 S BT BOTTLE
- PALNAT 100mg Caps 21 s BOTTLE
- Exhep Pen 300 Mg 3 Ml Enoxaparin Injection
- Rixilta 100 Mg Injection
- Eleftha 440 Mg Trastuzumab Injection
- Eleftha 375mg Trastuzumab Injection
- Abemaciclib 150 Mg Tablets
- Nintabid 150 Mg Capsules
- TIPANAT 20 BT OF 20 TABLETS BOTTLE
- Evaparin Enoxaparin Injection 40 Mg
- SUTINAT 25 28 CAPSULES BOTTLE
- LENVAT 4 3X10 CAPSULES BOTTLE
- BRACANAT 100 30 TABLETS BOTTLE
- AFANAT 30 28 S BT BOTTLE
- XPREZA 300mg TAB 14's BT BOTTLE
- BORTENAT 2MG INJ
- Ivermectin Cream 1.0% W W
- CAPNAT 500mg TAB 10 S STRIP
- LENALID 25MG CAPS 30 S BT BOTTLE
- PAZONAT 400 30 TABLETS BOTTLE
- SUTINAT 12.5 28 CAPSULES BOTTLE
- Ramiven Abemaciclib Tablets
- Aplet 60 Mg Tablet
- Ovucet Injection, Grade standard
- Geffon 250 Mg Tablets
- Pegasta Injection Peg filgrastim 6mg Pfs Intas
- Abretone Abiraterone Acetate 250 Mg Tablet
- THIOPLAN INJECTION 15MGVIAL
- ZOLDONAT 4 MG INJ - VIAL
- TORINAT 10 4X7 TABLETS BOTTLE
- LETRONAT 2.5MG TABLETS 10 s STRIP
- Mesacol Suppositories 500 Mg
- GEFTINAT 250 MG TABLETS 30 S BS STRIP
- PALNAT 75mg Caps 21 s BOTTLE
- Caditam 20 Mg Tablet
- Neukine Injection 300 Mg
- Sorafenat Tosylate Tablets
- Infections Drugs
- 1000mg Montaz Ceftriaxone And Tazobactum Injection
- 1.25gm Xone XP Ceftriaxone And Tazobactam Injection
- 250mg Montaz Ceftriaxone And Tazobactam Injection
- 500mg Montaz Ceftriaxone And Tazobactam Injection
- 2.5gm Tazim Ceftazidime And Avibactam Injection
- Itrazit Itraconazole Capsules
- 625mg Clavozit LB Tablets
- 500mg Ciprofloxacin Hydrochloride Tablets
- Disodium Hydrogen Citrate Syrup
- 500mg Xone Ceftriaxone Injection IP
- 100mg Mikacin Amikacin Sulphate Injection IP
- 500mg Mikacin Amikacin Sulphate Injection IP
- 500mg Azithral Azithromycin Tablets IP
- Xone SB Ceftriaxone And Sulbactam Injection
- 500mg Azitox Azithromycin Tablets
- 200mg Azithral Azithromycin Syrup
- 250mg Mikacin Amikacin Sulphate Injection
- 250mg Azithral Azithromycin Tablets IP
- 500mg Amitax Amikacin Sulphate Injection USP
- 1000mg Xone Ceftriaxone Injection IP
- 1.5gm Merosure SB Meropenem And Sulbactam Injection
- 1gm Merosure Meropenem Injection IP
- 125mg Xone Ceftriaxone Injection IP
- Lyopac G Lycopene Multivitamin Multimineral And Antioxidants Tablets
- Cefotaxime Sodium Injection IP
- 250mg Mox Amoxycillin Trihydrate Capsules IP
- 1000mg Tazid Ceftazidime Injection IP
- 500mg Taxim Cefotaxime Sodium Injection IP
- 1500mg Taximax Cefotaxime And Sulbactam Injection
- 250mg Amitax Amikacin Sulphate Injection USP
- 2.25mg Pipzo Piperacillin And Tazobactam Injection IP
- 200mg Podzit Cefpodoxime Proxetil Dispersible Tablets
- Pharmaceutical Medicines
- Dorsun T Eye Drops
- IVERMECTIN TABLETS 6MG
- PAZONAT 200 30 TABLETS BOTTLE
- Bendamustine Hydrochloride Injection
- Unigef 250 Mg Tablets
- RASBURNAT 1.5mg INJ VIAL
- Nintedanib Capsule 150 Mg
- Bortecad Bortezomib 2 Mg Injection
- 250mg Gefitinib Tablets
- Hertraz 440 Mg Trastuzumab Injection
- Borviz 2mg Anti Cancer Injection
- Posaconazole Tablet 300 Mg
- Mounjaro 5 Mg
- Nivolumab 40 Mg
- Vaxirab N Vaccine
- Vorier 200mg Tablet
- Meropenem 1gm Injection
- Desloratadine Tablet USP
- Veenat 400 Mg Imatinib Tablets
- 200mg Voritek Tablet
- Relilance Reliferon Interferon Alpha 2b Injection
- SORAFENAT 200 Mg TAB 30 S BT BOTTLE
- Tamoxifen 20mg Tablets
- Carmustine 100 Mg Injection
- Bicalutamide 50 Mg Tablet
- Artemether Injection 80 Mg
- TORINAT 5 4X7 TABLETS BOTTLE
- Alectinib Capsules 150 Mg
- Dorzolamide Cosopt Dorzox T Eye Drops
- Eye Drop
- Just Tear Eye Drop
- Paracaine Eye Drop
- Xinep Eye Drop
- 0.05% Hyclo Cyclosporine Eye Drops
- 0.9mg Cequa Cyclosporine Eye Drops
- Dexnova Eye Drop
- 0.1% Hyclo Cyclosporine Eye Drops
- Tizta Eye Drop
- 0.2% wv Patvira Olopatadine Hydrochloride Ophthalmic Solution
- 1mg Xlha Sodium Hyaluronate Ophthalmic Solution
- 0.5% Occumox Moxifloxacin Eye Drop IP
- Tropicacyl Eye Drop
- 0.03%wv Careprost Bimatoprost Ophthalmic Solution
- 1% wv Just Tear Liquigel Carboxy Methylcellulose Sodium Lubricant Eye Drops
- Zivifresh Gel Carboxymethylcellulose Eye Drops IP
- Carpinol Pilocarpine Nitrate Eye Drop IP
- Xiatra Travoprost Eye Drops IP
- Latocom Latanoprost And Timolol Ophthalmic Solution IP
- Hyvet 0.18% Sodium Hyaluronate
- Carboxymethylcellulose Eye Drops
- Latoprost Eye Drop
- 0.5%wv Zivimox LP Moxifloxacin And Loteprednol Etabonate Ophthalmic Solution
- 0.25%wv Alcatop Alcaftadine Ophthalmic Solution
- Blood Collection Tube
- Vacsure - Vacuum Blood Collection Plain Tubes (without additive)
- Vacsure - Vacuum Blood Collection EDTA K2 Tubes
- Vacsure - Vacuum Blood Collection EDTA K3 Tubes
- Vacsure - Vacuum Blood Collection Glucose (Fluoride-Oxalate) Tubes
- Vacsure - Blood Collection Sodium Citrate 3.2% Tubes (with aluminium pouch pack)
- Vacsure - Vacuum Blood Collection (Sodium Citrate 3.8% tubes (with aluminium pouch pack)
- Vacsure - Vacuum Blood Collection Lithium Heparin Tubes
- Vacsure - Blood Collection Sodium Heparin Tubes
- Vacsure - Vacuum Blood Collection EDTA K2 Tubes
- Vacsure - Vacuum Blood Collection EDTA K3 Tubes
- Vacsure - Blood Collection Sodium Heparin Tubes
- Vacsure - Vacuum Blood Collection Plain Tubes (without additive)
- Vacsure - Vacuum Blood Collection EDTA K3 Tubes
- Vacsure - Vacuum Blood Collection Glucose (Fluoride-Oxalate) Tubes
- Vacsure - Blood Collection Sodium Heparin Tubes
- Vacsure - Vacuum Blood Collection Clot Activator + Gel Tubes
- Vacsure - Vacuum Blood Collection Clot Activator + Gel Tubes
- Vacsure - Vacuum Blood Collection Clot Activator Tubes
- Anti Cancer Drugs
- TROMBONAT 50 TABLETS 7TAB 4 X 7 S STRIP
- Vivitra 440mg Injection Trastuzumab
- Nexavar Sorafenib 200 Mg Tablets
- VEENAT 100mg CAPS SHRINK WRAP 120 s BT BOTTLE
- Sunitinib 50 28 CAPSULES BOTTLE
- Onco Bcg Vaccine
- Dacarbazine 200mg Injection
- Doxorubicin Liposomal Injection
- Cytarabine 500mg Injection
- Aromita 1mg Tablet
- VEENAT 400 MG TABLETS 30 s BT BOTTLE
- Bortezomib 2.5 Mg
- Osimertinib Tablets 80 Mg
- Erlocip 150mg, 100mg Erlotinib Tablets
- Evertor10mg, 5mg Everolimus Tablets
- Tegonat 20 CAP 7 S Blister STRIP
- Anti Cancer Injection
- Vintor Erythropoietin Injection
- Herti Trastuzumab 440 Mg Injection
- Herti 440mg Injection
- CARFILNAT INJ 60MGVIAL
- Pertuzumab Sigrima 420mg Injection Zydus
- Cyphos 200mg Cyclophosphamide Injection
- INTAS Azacitidine 100 Mg Injection AZADINE
- TRABEC INF. 1MG VIAL
- Bortenat Bortezomib 2mg 3.5mg Injection
- Lorviqua Lorlatinib Lorbrena
- Kemocarb Injection 450 Mg
- KABANAT INJECTION 60MG 1.5ML
- NATDECITA INJECTION 50MGVIAL
- PEMNAT 100 MG INJ 1 S VIAL
- PEMNAT 500 MG INJ 1 S VIAL
- Anti Cancer Tablets
- CAZANAT 40 mg Tabs. 30 s BT BOTTLE
- REGONAT TABLETS 40 MG 28 S BT BOTTLE
- IDEENAT 100 30 TABLETS BOTTLE
- Nitib 140Mg Capsules
- LENALID 5 MG BT 30 S BOTTLE
- Geftinat 250mg Gefitinib Tablets IP
- IDEENAT 150 30 TABLETS BOTTLE
- KNILONAT 150MG CAPS 10 s BS STRIP
- Afatinib Afayro 20mg 40 Mg 30mg Tab 28 Cap Glenmark
- SORAFENAT 200mg TAB 120 S BT BOTTLE
- Face Wash
- Apple Cider Vinegar Face Wash
- Chamomile And White Tea Skin Revival Face Wash
- L Glutathione Kojic Acid Diapalmitate And Arbutin Face Wash
- 1% Kojic Acid Glutathione And C Complex Face Wash
- Ceramide Vitamin C And Glycerin Face Wash
- 1% Oat Vitamin F Vitamin B5 And Allantoin Deep Nourish Face Wash
- Coffee And Cocoa Face Wash
- Retinol Face Wash
- Fruit Brightening Face Wash
- Papaya Deep Cleanse Face Wash
- Salicylic Acid And Glycolic Acid Foaming Face Wash
- Vitamin B5 Saffron And Papaya Glow Bright Face Wash
- Fresh Neem Pimple Control Face Wash
- Oral Suspension
- Myo Inositol Folic Acid Vitamin D Sachet
- Dextrose Zinc Vitamin C Lactobacillus Electrolytes
- Collagen Peptide Glucosamine And Methylcobalamin Sachet
- Orthoboon Collagen Peptide Glucosamine And Vitamin C Sachet
- Collapure Collagen Peptide Magnesium N Acetyl And Glucosame Sachet
- Curcumin Longa Sachet
- Zenc Flora Prebiotic And Probiotic Sachet
- Vitamin D3 Oral Solution
- Arzigen L Arginine Zinc And Folic Acid Powder
- L Glutathione Sachet
- L Arginine Proanthocyanidin And DHA Sachets
- L Arginine Sachets Manufacturing
- Laboratory Chemical
- Ayurvedic Tablets
- Antibiotic Injection
- Mama Earth Honey And Malai Day Cream
- Vaccine Products
- Contact Us
Danyelza Injection 40 Mg
Danyelza Injection 40 Mg Specification
- Packaging Type
- Vial
- Indication
- Relapsed or Refractory Neuroblastoma
- Salt Composition
- Naxitamab-gqgk 40 mg
- Life Span
- 24 Months
- Pacakaging (Quantity Per Box)
- 1 Vial per Box
- Dosage Form
- Injection
- Brand Name
- Danyelza
- Drug Type
- Prescription
- Ingredients
- Naxitamab-gqgk
- Physical Form
- Injection
- Function
- Antibody Therapy
- Recommended For
- Neuroblastoma Patients
- Dosage
- 40 mg per vial
- Dosage Guidelines
- As Directed by Physician
- Suitable For
- Adults and Children
- Quantity
- 1 Vial
- Storage Instructions
- Keep In A Cool And A Dry Place
- Appearance
- Sterile Solution
- Box Contains
- Single Use Vial
- Clinical Use
- Oncology Therapy
- Therapeutic Area
- Pediatric Cancer
- Route of Administration
- Intravenous
- Prescription Status
- Rx Only
- Reconstitution Required
- Yes
Danyelza Injection 40 Mg Trade Information
- Minimum Order Quantity
- 1 Vial
- Supply Ability
- 5000 Vials Per Month
- Delivery Time
- 2-10 Days
About Danyelza Injection 40 Mg
Danyelza Injection 40 Mg stands as a best-seller and elite offering in the realm of pediatric oncology. Highlighted for its flawless formulation, this preeminent antibody therapy contains 40 mg of naxitamab-gqgk per sterile, single-use vial. Its majestic efficacy is tailored for patients battling relapsed or refractory neuroblastoma. Exclusively available on prescription, Danyelza is suitable for both adults and children, administered intravenously after reconstitution. It remains stable for up to 24 months under optimal, dry storage. Trusted by oncologists, Danyelza represents a remarkable innovation in pediatric cancer care.
Competitive Advantages & Key Features of Danyelza Injection 40 Mg
Danyelza Injection 40 Mg is acclaimed for its rapid action against neuroblastoma and exceptional safety profile. Its single-use, sterile vial ensures convenient handling and minimal wastage. As an elite monoclonal antibody therapy, Danyelza achieves robust response rates where traditional treatments falter. The flawless preparation process guarantees optimal potency, aligning with global quality benchmarks. Danyelza's pediatric focus, broad suitability, and easy intravenous administration mark it as a highlighted choice among advanced oncology solutions.
Certifications, Outlay, and Packaging Details for Danyelza Injection 40 Mg
Certified to meet preeminent pharmaceutical standards, Danyelza Injection is available from India's trusted suppliers, exporters, and wholesalers. The outlay covers fully compliant, secure goods transport and robust packaging-each vial preserved in a high-quality box to minimize potential expenditure due to spoilage. FOB port arrangements are flexible, tailored for efficient international shipping. Every 1-vial box is professionally labeled for accurate inventory management and seamless logistics from warehouse to healthcare provider.
Competitive Advantages & Key Features of Danyelza Injection 40 Mg
Danyelza Injection 40 Mg is acclaimed for its rapid action against neuroblastoma and exceptional safety profile. Its single-use, sterile vial ensures convenient handling and minimal wastage. As an elite monoclonal antibody therapy, Danyelza achieves robust response rates where traditional treatments falter. The flawless preparation process guarantees optimal potency, aligning with global quality benchmarks. Danyelza's pediatric focus, broad suitability, and easy intravenous administration mark it as a highlighted choice among advanced oncology solutions.
Certifications, Outlay, and Packaging Details for Danyelza Injection 40 Mg
Certified to meet preeminent pharmaceutical standards, Danyelza Injection is available from India's trusted suppliers, exporters, and wholesalers. The outlay covers fully compliant, secure goods transport and robust packaging-each vial preserved in a high-quality box to minimize potential expenditure due to spoilage. FOB port arrangements are flexible, tailored for efficient international shipping. Every 1-vial box is professionally labeled for accurate inventory management and seamless logistics from warehouse to healthcare provider.
FAQ's of Danyelza Injection 40 Mg:
Q: How is Danyelza Injection 40 Mg administered to patients?
A: Danyelza Injection 40 Mg must be reconstituted and then administered intravenously by healthcare professionals, following physician-directed dosage guidelines.Q: What is the primary clinical use of Danyelza Injection 40 Mg?
A: Danyelza Injection is primarily used for the treatment of relapsed or refractory neuroblastoma in both children and adults within oncology therapy.Q: When should Danyelza Injection be considered for neuroblastoma patients?
A: Danyelza is indicated for patients who have experienced relapse or have refractory neuroblastoma, typically following unsuccessful conventional treatments.Q: Where should Danyelza Injection 40 Mg be stored before use?
A: The vial should be kept in a cool, dry place as per storage instructions to maintain its stability and efficacy up to 24 months.Q: What is the recommended process for preparing Danyelza Injection 40 Mg?
A: Before administration, the injection requires reconstitution by a qualified healthcare professional, ensuring a sterile environment and accurate preparation.Q: How does Danyelza Injection 40 Mg benefit neuroblastoma patients compared to other therapies?
A: As a targeted antibody therapy, Danyelza offers enhanced effectiveness and safety in treating neuroblastoma, especially for patients who have not responded optimally to previous treatments.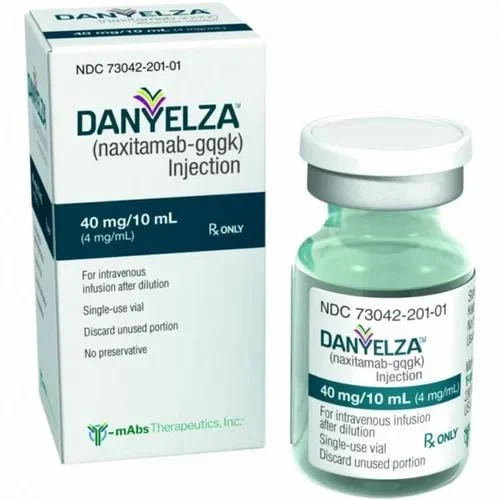
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmaceutical Injection Category
Azacitidine 100 Mg Injection
Price 3707.81 INR / Vial
Minimum Order Quantity : 1 Vial
Drug Type : Other, Allopathic
Origin of Medicine : India
Storage Instructions : Store below 30C
Physical Form : Other, Injection
Bortecad 2.5mg Bortezomib Injection
Price 3180 INR / Vial
Minimum Order Quantity : 1 Vial
Drug Type : Other, Allopathic
Origin of Medicine : India
Storage Instructions : Store below 25C
Physical Form : Other, Injection
Hynidase Inj
Price 172.52 INR / Box
Minimum Order Quantity : 10 Boxes
Drug Type : Injection
Origin of Medicine : India
Storage Instructions : Cool & Dry Place
Physical Form : Liquid
Lantus Cartridge
Price 2691.49 INR / Box
Minimum Order Quantity : 10 Boxes
Drug Type : Injection
Origin of Medicine : India
Storage Instructions : Cool & Dry Place
Physical Form : Liquid





 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese